The U.S. Food and Drug Administration approved Wednesday a breakthrough cancer drug to treat childhood leukemia. The “living drug” consists of genetically edited cells from a patient’s immune system, which are reinserted into the patient to attack and kill leukemia cells.
The drug, manufactured by Novartis AG, received approval from an FDA advisory committee in July and was definitely approved on Wednesday, according to a statement from the FDA.

The drug, known as Kymriah (tisagenlecleucel), will be used to treat certain pediatric and young adult patients with a form of acute lymphoblastic leukemia (ALL).
CAR-T therapy has already saved the lives of dozens of patients
Kymriah was approved in the United States for the treatment of patients up to 25 years of age with B-cell precursor ALL, according to the federal health agency.
“We’re entering a new frontier in medical innovation with the ability to reprogram a patient’s own cells to attack a deadly cancer,” said FDA Commissioner Scott Gottlieb. “New technologies such as gene and cell therapies hold out the potential to transform medicine and create an inflection point in our ability to treat and even cure many intractable illnesses.”
The drug is a chimeric antigen receptor T-cell or CAR-T, which takes immune system cells from a patient’s body, modifies its genes, and then the “upgraded” T-cells are placed back into the patient. The modified genes can track down and kill cancer cells effectively.
Novartis Pharmaceuticals announced the price for its one-time infusion would be $475,000, but noted there would be no charge for patients who didn’t show a response within a month of receiving the treatment.
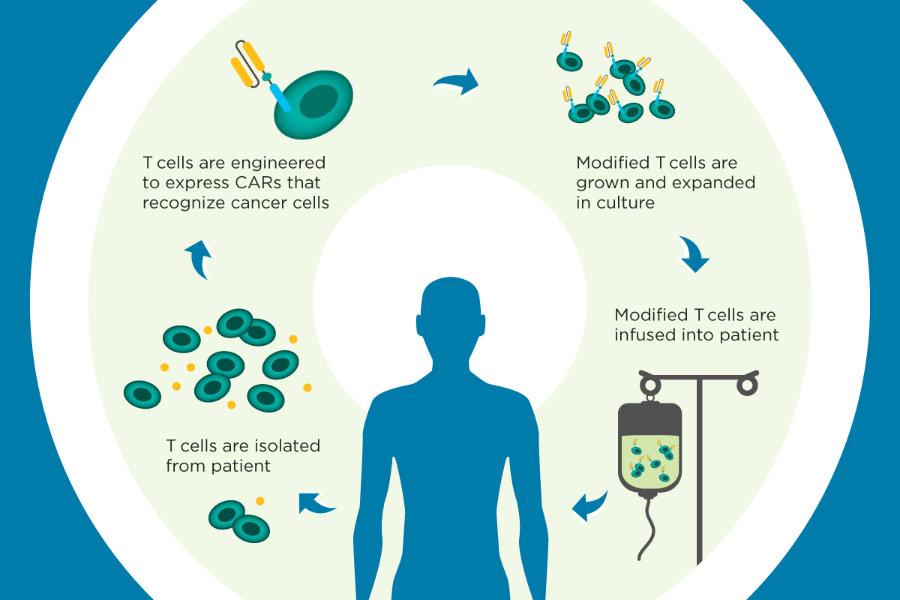
“This is a brand new way of treating cancer,” Dr. Stephen Grupp of Children’s Hospital of Philadelphia, who treated the first kid with CAR-T cell therapy, told ABC News. “That’s enormously exciting.”
The child Grupp treated was a girl who had been near death but now is cancer-free for five years. Other patients who have received Kymriah have also seen their cancer disappear, such as Austin Schuetz, who in 2013 (at the age of 6) received the Novartis drug and currently shows no signs of leukemia or any other type of cancer.
New FDA-approved ‘living drug’ will cost $475,000 for a one-time dose
While some patients receiving CAR-T cell therapy have relapsed after their treatments, is a small percentage, and doctors are confident this new type of treatment is the new frontier to treat cancer. However, some of the side effects from this treatment can even be life-threatening.
An immune overreaction known as “cytokine release syndrome” can cause high fevers, dropping blood pressure and in some severe cases organ damage. The FDA designated an alternative treatment for those side effects on Wednesday. Some experts have shared some concerns regarding the new technology, as sometimes leukemia can develop resistance, or patients can worsen while waiting for the new cells.
The FDA said the CAR-T therapy will only be available (for now) in medical centers specially trained and certified to handle the treatment. Novartis estimates there are about 32 centers around the country suited to manage the technology and said at least 20 facilities could begin offering care within the next month.
Novartis said the company is working with the Medicaid program and some private insurers and expects broad coverage. The company will also offer financial assistance for some cases, but they didn’t promise all patients will be able to get the therapy. While the $475,000 price can seem too expensive, some bone marrow transplants (the alternative – which CAR-T therapy seeks to replace) can cost more than half a million dollars, according to Grupp, who led a study on the drug for Novartis.
“I don’t want to be an apologist for high drug prices in the U.S.,” said Grupp, according to ABC News. But if it’s the last treatment patients need, “that’s a really significant one-time investment in their wellness, especially in kids who have a whole lifetime ahead of them.”
Source: ABC News
