The U.S. Food and Drug Administration has just approved Brineura, the first medical treatment for the rare CLN2 disease, or what it’s known as Batten disease which affects mainly kids between 2 and 4 years old. This would be one of the most expensive treatments the FDA has approved, costing a total of $700,000 per year
Brineura is an enzyme replacement therapy. Its active ingredient is Cerliponase alfa, a laboratory-developed form of the human enzyme TPP1, which is deficient in people with the CLN2 disease.
“The FDA is committed to approving new and innovative therapies for patients with rare diseases, particularly where there are no approved treatment options,” said Julie Beitz of FDA’s Center for Drug Evaluation and Research. “Approving the first drug for the treatment of this form of Batten disease is an important advance for patients suffering from this condition.”
Batten Disease affects about 4 in every 100,000 births in the United States
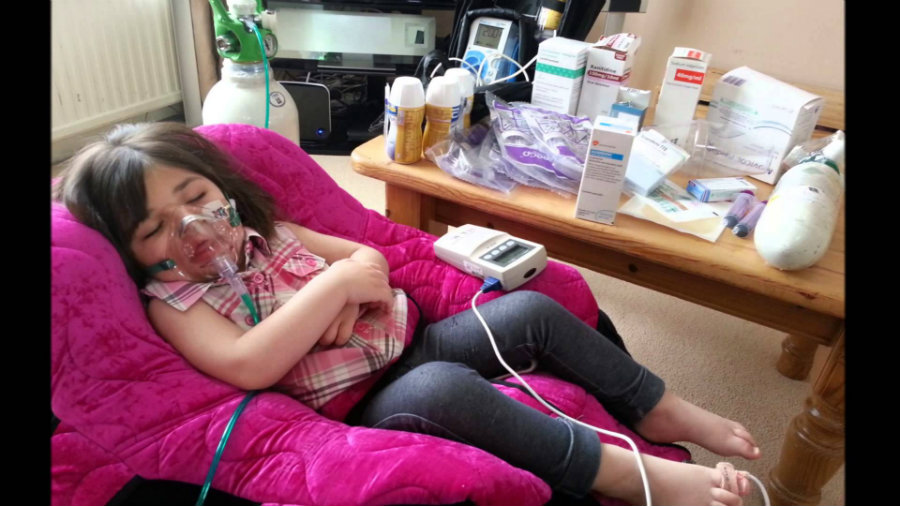
CLN2 is a one of a group of diseases known as neuronal ceroid lipofuscinoses (NCLs), or collectively called Batten disease. It was first described a century ago. CLN2 is a rare inherited condition that affects the nervous system. This disease causes language delay, difficulty in coordinating movements or ataxia, and epilepsy. Batten disease affects mainly the motor skills. Consequently, people with this condition have difficulties walking and sitting. According to the FDA, in the late infantile form of the disease, symptoms usually begin between ages 2 and 4.
Children with Batten Disease develop muscle twitches (myoclonus), and they eventually lose vision. Patients tend to require a wheelchair by late childhood, and the children usually die when they are teens. According to the FDA, in the United States, there are 2 to 4 cases of Batten disease in every 100,000 births.
FDA gives hope to patients with Batten disease
With the FDA’s announcement, patients of CLN2 and their families will have a little bit of hope, since it is the first approved treatment for this disease. It is called Brineura (cerliponase alfa). It would slow the loss of ability to walk in patients with ages 3 and older with late infantile neuronal ceroid lipofuscinosis type 2 (CLN2), also known as tripeptidyl peptidase-1 (TPP1) deficiency.
The treatment must be applied by a professional since it needs to be performed under sterile conditions to minimize the risks of infection, and the enzyme replacement drug needs to be applied directly into the cerebrospinal fluid using a surgically implanted reservoir and a catheter in the head (intraventricular access device). The administration of the entire Brineaura infusion lasts approximately 4.5 hours. Additionally, the FDA stated that pretreatment of patients – using antihistamines with or without antipyretics (drugs for prevention or treatment of fever) or corticosteroids – must be done about an hour before the start of the infusion.
The efficacy of Brineura was approved in a non-randomized, single-arm dose escalation clinical study which included 22 children with CLN2. All of them were at least 3 years old, and they had motor and language problems.
Side effects of Brineaura
According to a clinical study, that kept track on 24 children with CLN2 whose ages ranged between 3 and 8, the most common side effects of the treatment included fever, vomiting, slow heart rate (bradycardia), heart problems, seizures, increased CSF white blood cell count (pleocytosis), decrease or increase in CSF protein, headache, and irritability.
The drug has not been approved for patients younger than 3 years old. In order to be approved for younger patients, the FDA requested the manufacturer to carry out a clinical study that lasts at least 10 years, so the long-term effects of the treatment are sizable
Brineura should not be administered to patients if there are symptoms of acute intraventricular access device-related complications, such as leakage or device failures. If that is the case, the professional administrator should discontinue the infusion of the drug. Health cares should observe vital signs before infusion treatment. They should also carry out electrocardiogram (ECG) monitoring during infusion in patients with a history of slow heart rate.
Brineura treatment will cost $702,000 per year
Though the approval of Brineura for the treatment of CLN2 patients is a tremendous advance for the patients and the sake of science, it represents an enormous cost too. Every biweekly infusion of the treatment has a price of $27,000 which is equivalent to $702,000 per year, according to BioMarin, which is the manufacturer of the drug.
“Treating children with CLN2 disease requires an extraordinary amount of collaboration between families, hospitals, advocates, and physicians. We are grateful for the partnership of all those involved and look forward to continuing to work together to make Brineura accessible to children who may benefit,” saidJean-Jacques Bienaimé, chairman and chief executive officer of BioMarin.
The FDA granted Brineura a priority review and a Breakthrough Therapy designations. The drug also received Orphan Drug designation, which increases incentives to assist in the development of treatment for rare and terminal diseases.
Source: U.S. Food and Drug Administration
