Bayer’s new contraceptive device Kyleena is now approved in the U.S. The FDA gave the green light to the levonorgestrel-releasing system to be sold in the United States as another option for women that want to avoid pregnancy or regulate their cycle with hormones. Kyleena will be available in October 2016.
Kyleena is an intrauterine device made of flexible plastic with a T shape that contains 19.5 mg of levonorgestrel. The hormone is slowly released into the woman’s uterus. Levonorgestrel stops the womb lining from thickening, which prevents pregnancy and also reduces monthly bleeding. The new device is a long-acting reversible contraceptive (LARC), a category of potent contraceptives that has returned to the market.
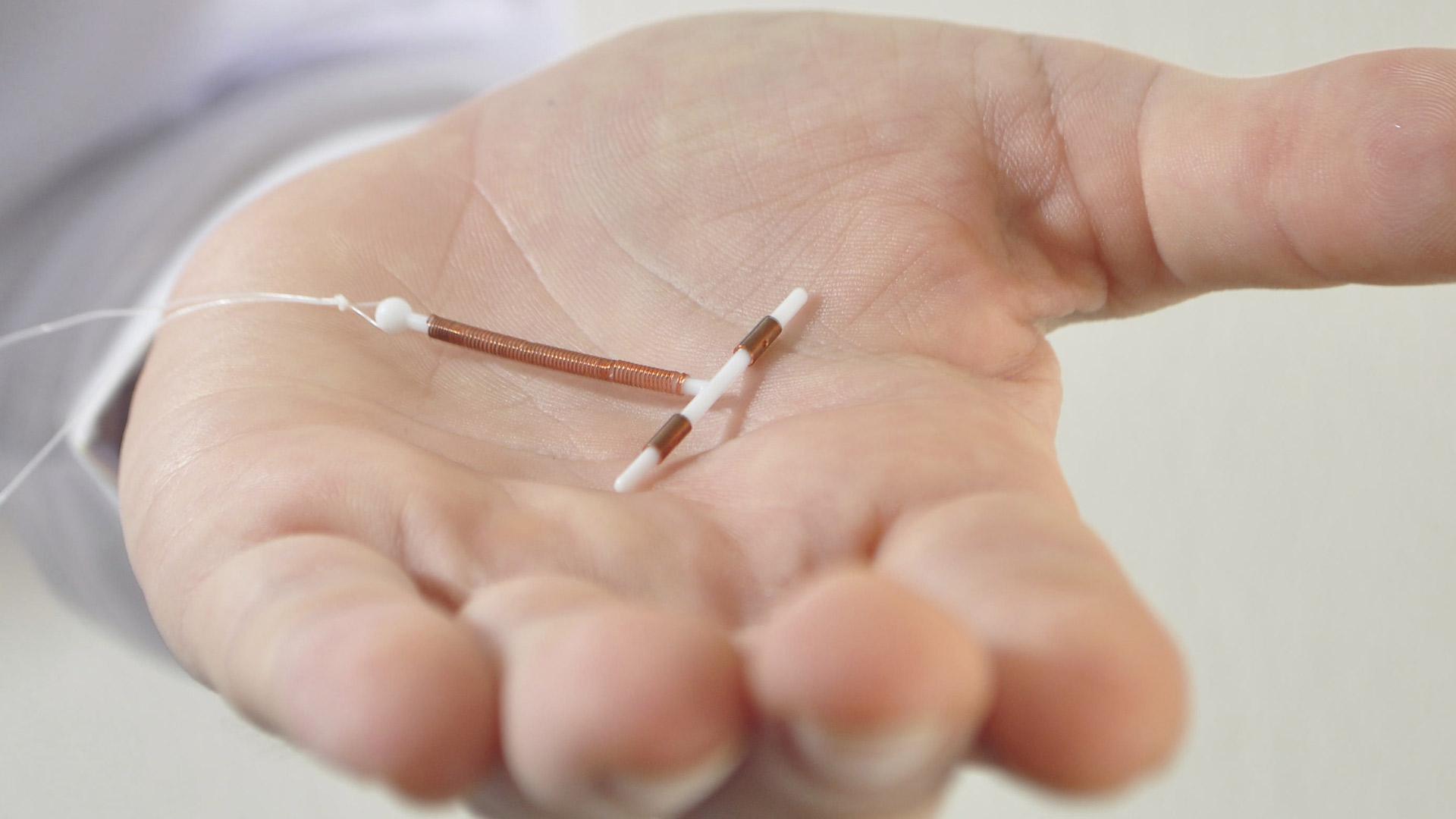
Kyleena lasts five years, and it is similar to other LARCs. It is almost as effective as sterilization, according to the U.S. Centers for Disease Control and Prevention.
LARCs are more efficient than other contraceptive methods, including pills and patches. Bayer’s recently-approved IUD will be on drugstores in October and would be purchased by prescription.
A woman using Kyleena will experience fewer days of bleeding, and for some, the period and spotting will stop altogether, according to Bayer. The recently approved device contains progestin, which reduces a woman’s bleeding.
The IUD is placed in the uterus by a health provider during an in-office visit. It may also be removed by a health provider anytime.
Anita L. Nelson, MD, professor and chair of Obstetrics and Gynecology at Western University of Health Sciences, said in a press release that Kyleena is highly effective at preventing pregnancy.
Professor Nelson added that device might be an appropriate choice for women who want a low-dose non-daily birth control method.
Clinical trials behind Kyleena’s approval
To establish Kyleena’s efficacy in preventing pregnancy, a clinical trial involving 1452 women was performed among subjects aged 18 to 35 years, who all received the IUD.
The trial excluded women with fewer than six weeks postpartum, and those with a long history of ectopic pregnancy. Women with a clinically significant ovarian cyst or HIV and women at high risk for sexually transmitted infections were also excluded.
Kyleena’s contraceptive reliability was assessed using the Pearl Index, a calculator that measures the effectiveness of birth control methods. This index was calculated based on 28-day equivalent exposure cycles.
Bayer’s IUD proved to be effective and showed that women who want to get pregnant just have to remove the device and wait for a few months for the body to adapt to its regular hormonal dose.
71 percent of 163 women who desired pregnancy after the trial conceived within 12 months after having the small T-shaped piece removed. Kyleena is not Bayer’s first hormonal IUD. The German pharmaceutical also offers Mirena and Skyla.
Source: Reuters
